Washington State University researchers have received a National Science Foundation grant to better understand and design catalytic reactions that remove harmful pollutants in automobile catalytic converters and other emissions control systems.
The work could someday lead to the development of more efficient catalysts, reducing the amount of expensive precious metals needed to convert a car’s deadly carbon monoxide emissions to safer carbon dioxide.
Led by Jean-Sabin McEwen, professor in the Gene and Linda Voiland School of Chemical Engineering and Bioengineering, the $300,000, Early-concept Grants for Exploratory Research (EAGER) grant provides support for this high-risk, high-impact work.
For generations, catalysts have often been developed by simply seeing what works, relying largely on past experience and accepted practices, said McEwen, who also holds a joint appointment at Pacific Northwest National Laboratory. Researchers often design catalysts with only limited understanding of their underlying mechanisms.
“How can you improve the catalyst in the first place if you don’t really understand how it behaves,” he said. “You have to know how the catalyst restructures.”
As engines have become more efficient in recent years, their combustion temperature has become lower, making it harder for catalytic converters to work and creating, paradoxically, more harmful emissions. At the same time, researchers and the auto industry would like to reduce the amount of expensive precious metals, like platinum, needed to capture and bind the harmful pollutants.
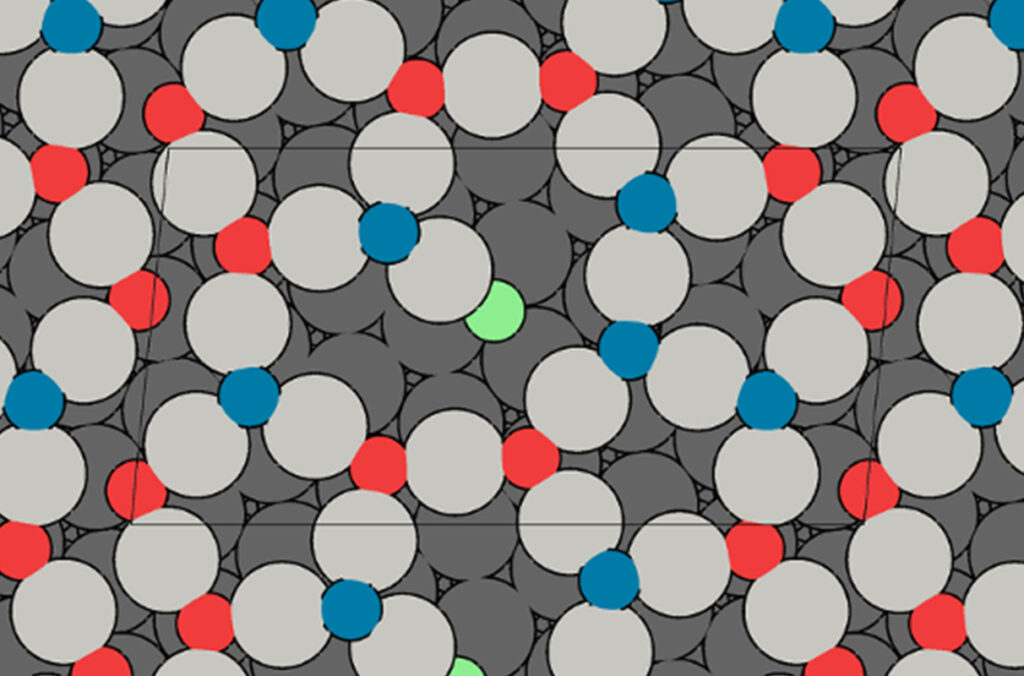
In this project, the researchers are investigating how a thin layer of copper oxide undergoes restructuring when combined with atomically dispersed metals such as platinum — a combination showing promise for controlling vehicle exhaust emissions at lower temperatures. A few years ago, a WSU-led team collaborated with experimentalists at Tufts University to develop a similar catalyst capable of converting carbon monoxide at relatively low temperatures of around 150 degrees Celsius.
In that work, they thought the oxide structure was stable as it fed oxygen to the reaction to convert carbon monoxide to carbon dioxide. Recently, using the latest tools of machine learning, they learned that the structure transforms “to a whole zoo of other structures,” said McEwen.
“This oxide structure is very dynamic and changes completely,” he said. “We couldn’t even imagine what all these structures could be.”
To better understand and improve the reaction, the researchers are using a powerful computer tool developed at Aarhus University in Denmark. This program works with the lab’s existing software to identify the most stable arrangements of atoms on catalytic surfaces. By combining advanced search algorithms with traditional methods, the researchers can explore a wide range of possible surface structures — something that would be extremely difficult to do using standard techniques alone.
“These corresponding structures are very dynamic, and looking at their dynamicity has often not really been considered until recently, but that’s what’s really happening,” McEwen said. “Taking into account the full complexity of these corresponding structures is really why this effort is so important.”
“We’re very excited about doing this research, revisiting some of the results, and arriving at a more complete model,” he added.
In addition to McEwen, collaborators on the project include researchers at Tufts University, Aarhus University in Denmark and the University of Nottingham in the United Kingdom.