If used in the right way, water could help improve catalysts used for thermochemical reactions: lowering energy barriers and more efficiently converting biomaterials to fuels, according to a new study in the journal, Catalysis Today.
Although it is generally preferable to remove water from reactions, work led by Jean-Sabin McEwen and Di Wu in the Gene and Linda Voiland School of Chemical Engineering and Bioengineering, could lead to better ways of using water in processes that make renewable energy more viable.
“Water could actively participate in the reaction to lower energy barriers and energy costs,” said McEwen.
Many industries rely on catalysts for important reactions, including the fuels industry, for conversion of chemicals. But, industries have traditionally relied on intuition in many cases to improve their reactions – developing a “witch’s brew” to see what works.
“It’s why it is really important to understand these catalysts from a fundamental point of view, so that we can make informed, rational decisions instead of trying this and that,” said McEwen.

Biomass is the largest source for renewable energy, but one of the big challenges in using it as liquid fuel, or biofuel, is the large amount of oxygen it contains. Unless that oxygen is carefully removed, it can corrode engines and wreak havoc when mixed with fossil fuels. But removing it is cumbersome and expensive. To convert biomass to useable fuels similar to gasoline requires an extra, energy-intensive refining step to break the bonds between carbon and oxygen.
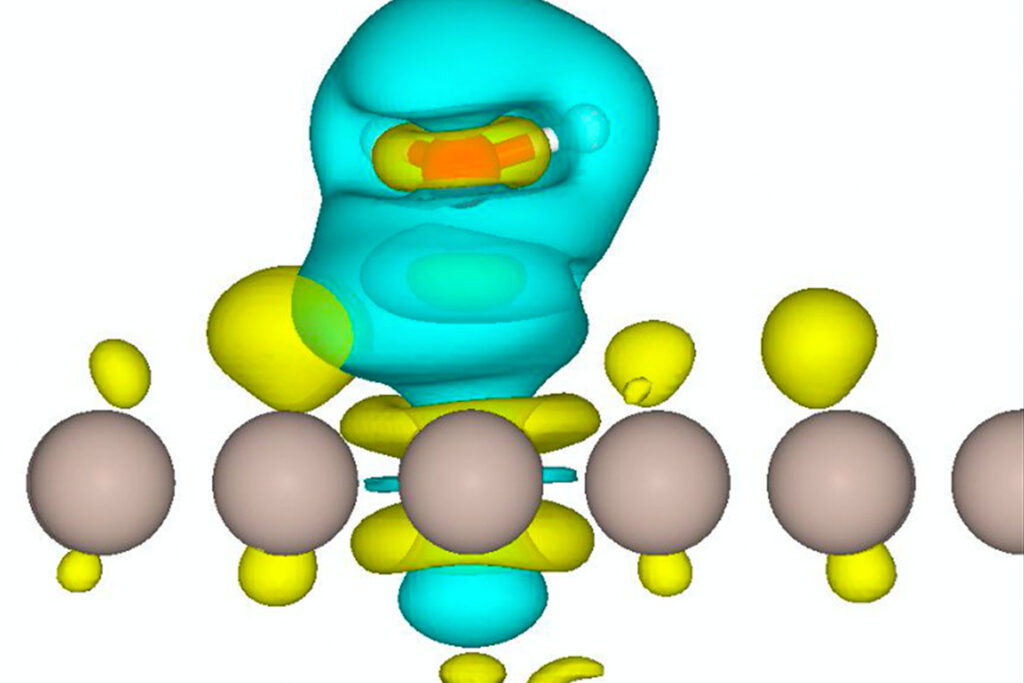
In their work, the researchers took a modeling and experimental thermodynamic approach to understand the fundamentals of the interaction energetics between water and a metal-carbon catalyst that is commonly tailored to optimize biofuels conversion.
The researchers initially tried looking at a model of a single water molecule and its binding strength to the metal-carbon catalyst. But, when they compared their model to calorimetric measurements, the numbers didn’t add up. They looked at the problem again and found that increasing the surface water population led to an enhanced affinity for more water. In other words, lots of water on the catalyst made for a stronger hydrogen bonds at the surface of the catalyst which, in turn, led to the production of more desirable fuels that contained less oxygen.

They also found new and unique pathways for water to aid in the conversion, serving as a shuttle of protons to the oils as elaborated in two parallel papers that were recently published in ACS Catalysis and Journal of Catalysis.
“A lot of previous work didn’t take into account the role of water in such reactions,” said McEwen.
While the work could lead to better ways of making use of water in reactions, it also could build knowledge for catalyst creators on why some of their catalysts simply fail and stop working. While they haven’t been looked at closely, water molecules are nearly always present in important reactions because the molecules are simply always in humid air.
“Fundamental knowledge is essential to understand the roles that water can play in the synthesis, activation, and regeneration of the catalysts,” he said.
The work was funded by the U.S. Department of Energy, Office of Basic Energy Sciences, Division of Chemical Sciences, Geosciences, and Biosciences, within the Catalysis Science program.