A team of Washington State and Tufts University researchers have uncovered fundamentals in how mirror-image molecules interact, a discovery that could have implications in biochemistry, medicine, and industry.
Chirality, the type of mirror symmetry that is seen in left and right hands, is seen throughout the universe – down to the molecular level. Hands and other chiral elements are mirror images of each other, but they don’t line up when one is placed on top of the other.
Chirality is critically important in designing chemicals for pharmaceuticals and other industries.Because many of the chemicals in organisms are chiral, drug molecules of the wrong “handedness” can affect people by creating enantiospecific, or non-comparable, interactions between a chiral drug molecule and biomolecules in the body. The first disastrous example of this type of interaction was thalidomide, a drug that was given to pregnant women in the early 1960s to alleviate morning sickness. The wrong handed version of the drug molecule reacted badly with the human biomolecules and led to numerous, severe birth defects and miscarriages.
In efforts to develop and improve catalysts and chemicals, such enantioselectivity is “often subtle and difficult to control catalytically,” and it breaks the symmetry of the system, said Jean-Sabin McEwen, associate professor in WSU’s Gene and Linda Voiland School of Chemical Engineering and Bioengineering.

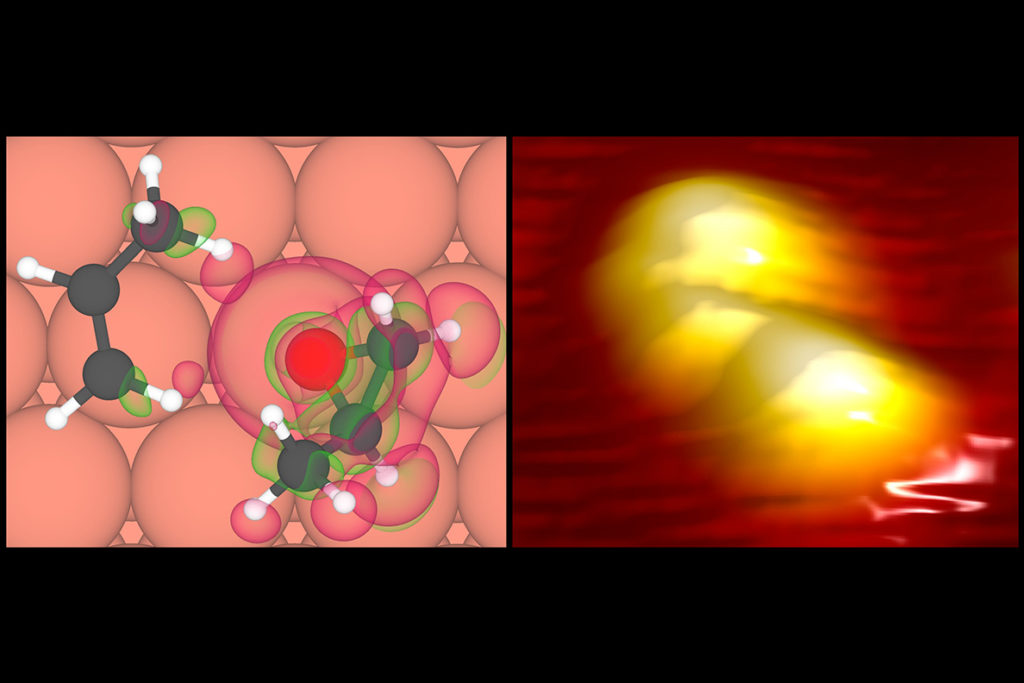
In the new study, published in the journal ACS Nano, the researchers, including McEwen and Charles Sykes, chemistry professor at Tufts University, studied the interaction of an individual chiral molecule with a molecule that became chiral when it landed on a surface.
Using high-resolution scanning tunneling microscopy, the team directly visualized the handedness of each individual molecule on the surface. They discovered that the chiral molecule docked more often with a certain chirality of its neighbor. They then quantified the interactions between the molecules, so as to shed light into this docking preference.
The symmetry breaking events that they saw in their model system are the origin of the enantiospecific interactions between a chiral drug molecule and chiral biomolecules, said McEwen.
“The work is of importance to the heterogeneous catalysis industry as it seeks ways to make just one chiral version of, for example, a drug molecule,” he said.
The work at Tufts and WSU was funded by the National Science Foundation with support from the Department of Energy’s Office of Biological and Environmental Research at Pacific Northwest National Laboratory.
Media Contact:
- Jean-Sabin McEwen, associate professor, Gene and Linda Voiland School of Chemical Engineering and Bioengineering, 509‑335‑8580, js.mcewen@wsu.edu
- Tina Hilding, communications director, Voiland College of Engineering and Architecture, 509‑335‑5095, thilding@wsu.edu